The development of a bioassay is, in essence, a long series of choices. An assay which reaches commercial use is the end of a single path through a near-infinite forest of decision points and optimisations. The eventual goal is to produce an assay which is as accurate and precise as possible, while still being practical to implement on a day-to-day basis. A key stage of this decision tree is the setting of the plate layout for the assay. Far from a simple question of convenience, an optimised plate layout needs to balance practicality, assay performance, and throughput. Here, we’ll examine the key influences on these decisions, and discuss the statistical benefits of a well-designed plate layout.
What is a Plate Layout?
Most bioassays utilise a microplate. These are trays – typically made of plastic – with a series of wells to contain reagent and product. To perform a bioassay, samples are loaded into the wells, a reaction is allowed to happen, which is then read by a plate reader to provide numerical results ready for statistical analysis.
Key Takeaways
- Plate layout directly impacts assay quality. The positioning of samples on a microplate can introduce systematic bias due to plate effects (e.g. edge, row, or column effects), influencing both accuracy and precision.
- Randomisation helps reduce bias but introduces practical challenges. Fully randomised layouts minimise location-based bias, but can be difficult to implement manually and may reduce throughput unless automation is used.
- Strategic layouts informed by uniformity testing can optimise performance. When plate effects are consistent and well understood, structured layouts (e.g. mirrored or blocked designs) can effectively minimise bias while maintaining practicality.
These microplates come in several forms, with the depth and shape of well and plate colour among the properties which can have an impact on the behaviour of the assay, but a key distinguishing factor is the number of wells on a plate. The plate which is most commonly used is the 96-well plate (8 rows by 12 columns), but 384-well (16Rx24C) and 1536-well (32Rx48C) plates are also used for some applications.
The plate layout, then, is the choice of where each dilution of each sample is placed on the microplate for the assay. Considerations may include how many samples are included, their physical location on the plate, and their proximity to other dilutions of the same or different samples. The plate layout can affect the accuracy and precision of the assay, as well as having an effect on the practicality and throughput of the assay.
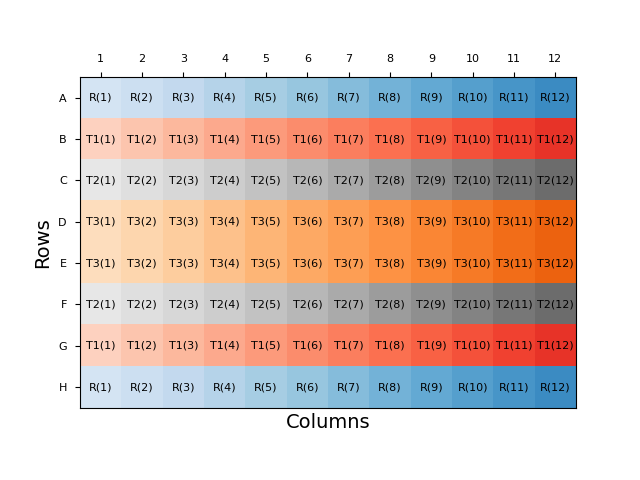
Throughout this blog, we will examine plate layouts based on a 96-well plate with four samples: a reference sample (R), and three test samples (T1-T3) each in duplicate. An example of a naïve plate layout, with the sample duplicates blocked together, is shown below:
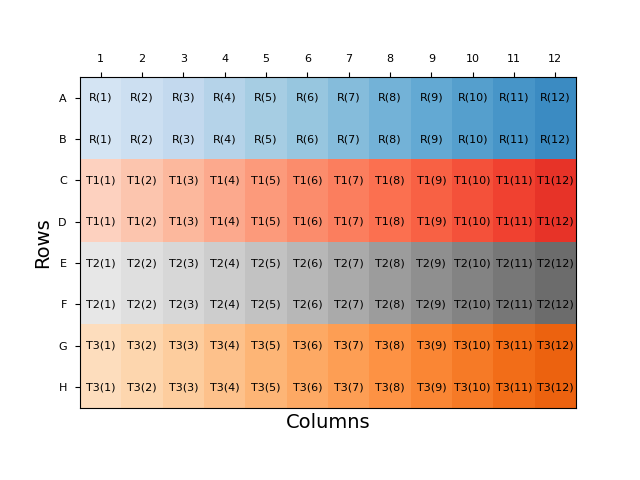
Each sample is represented by a different colour on the plate, with the intensity of the shading representing the dose applied to that well. Dose information is also included in brackets in the label for each cell. This representation will be used for all layouts discussed here.
Performance, Practicality, and Throughput
So, we have established that the three key considerations when designing a plate layout are:
- Performance: the ability of the assay to provide accurate and precise results.
- Practicality: the ease of performing the assay for an analyst.
- Throughput: how many samples can be analysed in a given period
Among these, the paramount concern is the assay’s performance. The presiding goal of an assay development process is to produce an assay which is fit for purpose, and an assay which cannot meet the accuracy and precision requirements will never succeed in that goal. The placement of samples on the plate can have a considerable impact on the assay’s performance.
Specifically, the measured response of an assay can be subject to plate or location effects. In an ideal world, a plate would be able to provide a consistent background against which the assay responses are measured. Realistically, however, responses from samples on different parts of the plate might be influenced by environmental conditions and the properties of the plate itself, which can lead to biases in the results. Perhaps the most commonly observed of these is the edge effect, where differences in the environment between wells at the edge of a plate and those nearer the centre can lead to biased responses. Indeed, edge effects can be so pervasive that it has become common practice to avoid using the outer wells of a plate in many cases.
Edge effects are not the only plate effect which can be of consequence. Even small variations in conditions across the plate can lead to gradients in responses, which are known as row or column effects. For example, if samples are applied to the plate by an analyst from left to right, it might be the case that fractionally less sample is applied in wells at the right end of a row than in those at the left end due to the dispensing properties of the pipette. Or, there might be a small temperature gradient across the plate during incubation due to the placement of the plate in the incubator. While steps should be taken to mitigate these effects at source, layout design can also be used to account for these biases.
It should also be noted that larger plates are more prone to location effects. This can be a consequence of the requirement of fitting more wells onto the plate. For example, smaller wells hold a smaller volume of reagents, meaning any biases as a result of pipetting can be amplified. The smaller well volume also means that any evaporation forms a larger proportion of the total volume of the well, which can amplify, for example, edge effects.
While maximising the performance of the assay is the primary goal of the assay development process, it is also important to consider both the practicality of performing the assay and the throughput of analysed samples which can be achieved. These qualities are not entirely independent: an assay which is easy for an analyst to perform is likely to achieve a higher throughput than a more complex assay, all else being equal. There are, nevertheless, trade-offs between the practicality and throughput, as well as with the assay’s performance.
Randomised Plate Layouts
In statistics, we typically control for environmental effects by randomising experimental units. A farmer testing fertilisers, for example, might randomise which product each field receives over the course of several experiments. This reduces the likelihood of biases being introduced into the results due to the properties of the different fields – one field may get more sunlight, or another might have more acidic soil. By randomising, the effect of each fertiliser on crop growth can be better isolated from the influence of the field environment.
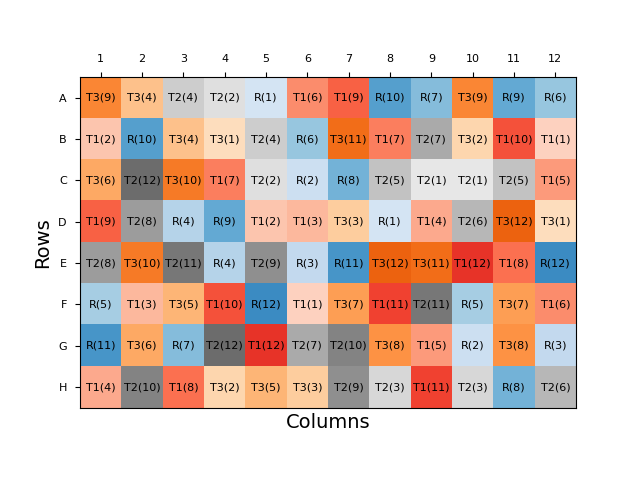
Similarly, we can randomise the location of sample dilutions on a bioassay plate to reduce the influence of location effects. From a performance point of view, the optimal form of this would be for the plate layout to be completely randomised: each sample dilution would be randomly assigned to a well for each analysis. An example of a fully randomised plate layout is shown below.
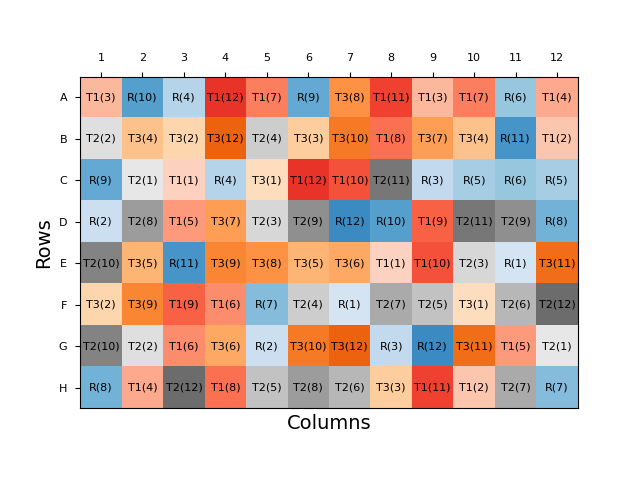
This reduces the likelihood of the results for a certain sample being biased by a plate effect as the doses of the sample are distributed across the plate.
From a practicality point of view, however, this is far from ideal. A plate layout with no patterns would be a nightmare for an analyst to prepare, particularly as the layout will necessarily change for every assay. Plating the analysis would be slow, and there would likely be many mistakes and rejected plates. Throughput would also, therefore, be greatly reduced.
Other plate layout approaches which combine more analyst-friendly patterns with some degree of randomisation include split-plot and strip-plot layouts. In the former, each sample replicate is randomly assigned a row, before the doses within each row are randomised. In a strip-plot design, the rows are again randomised, but the order of doses is randomised only once and is identical between each row. While these semi-random layouts are easier for an analyst to apply, they will still change between assays and, therefore, are less practical and can result in reduced throughput compared to an assay with a fixed plate layout. Automation can mitigate these issues, however: robotic dosing systems can be programmed to apply a randomised plate layout, removing the practicality dilemma.
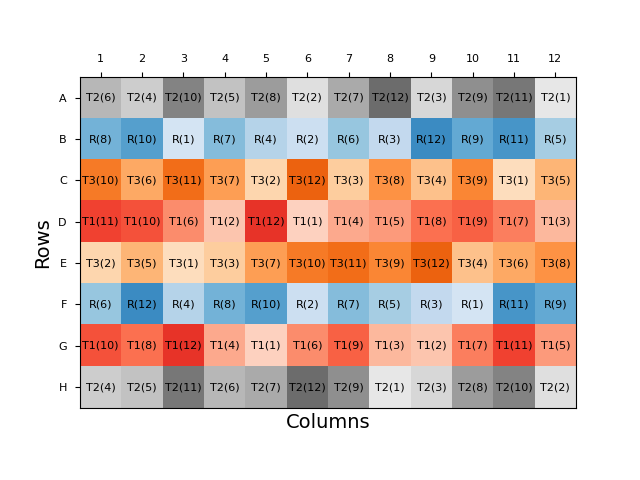
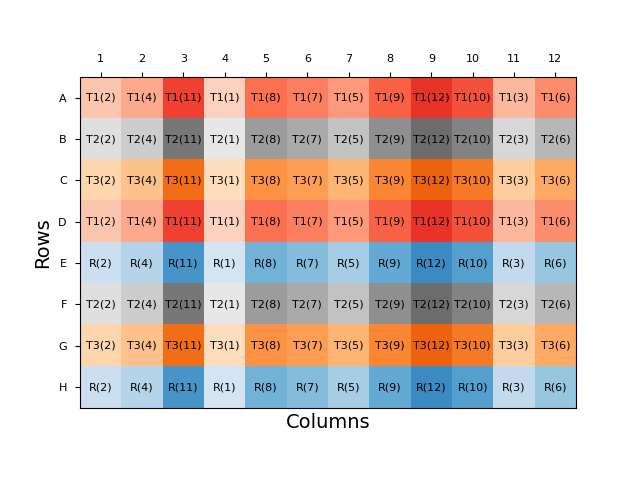
Uniformity Testing and Strategic Layout Choices
Randomised designs can be effective at reducing biases caused by plate effects, but they may not always provide the optimal approach. In cases where plate effects are known and are consistent between assays, we can make strategic layout choices to minimise their effect on results and maximise the performance of the assay.
So, how do we test for the presence of plate effects? A key tool in our arsenal is the uniformity trial. This is an assay in which all cells on a plate are assigned the same dose of the same sample. This is typically chosen to be a middle dose of the reference standard.
A middle dose is chosen as these are expected to fall in the linear portion of the sigmoidal dose-response curve. The response is changing rapidly with dose in this region of the curve, meaning small changes in the dose or environment will lead to a large change in the response. This means the experiment is more sensitive to influences on the response than if a dose which fell on an asymptote was chosen, for example.
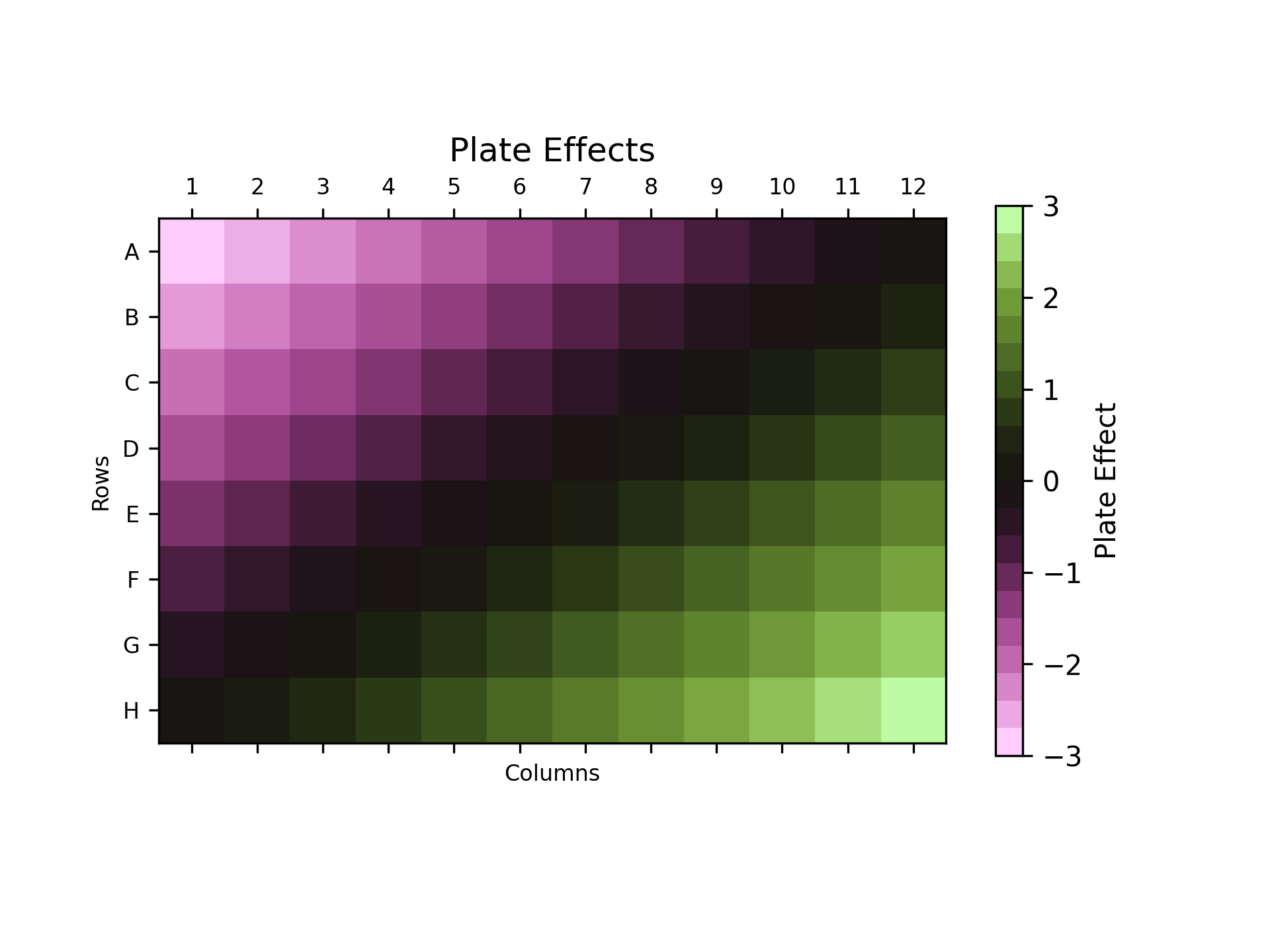
The plot above shows an example of results from a simulated uniformity trial. It shows the measured plate effect in each cell of a 96 well plate, with purple colours representing negative plate effects, greens representing positive plate effects, and blacks representing plate effects which are close to zero. The plate effects are defined by the difference between the expected response from the chosen dose and the response measured in the uniformity trial.
We can see that there is a clear trend here: the plate effect is negative in the top left of the plate, and increases smoothly to positive values in the bottom right. In an ideal world, the source of these plate effects could be identified and removed, but this is not always possible. Below we see the performance of the plate layouts we’ve discussed so far under the presence of these plate effects.
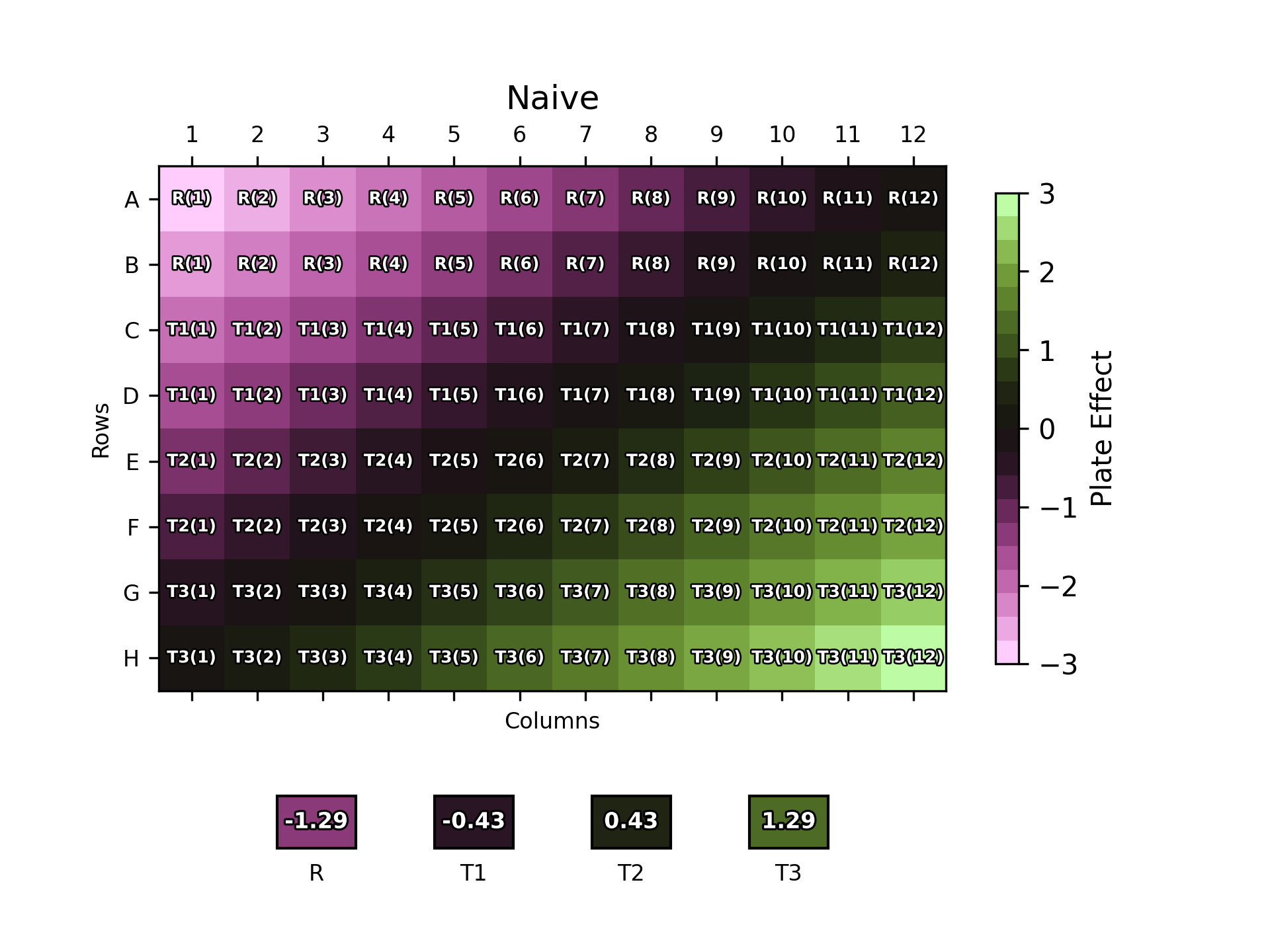
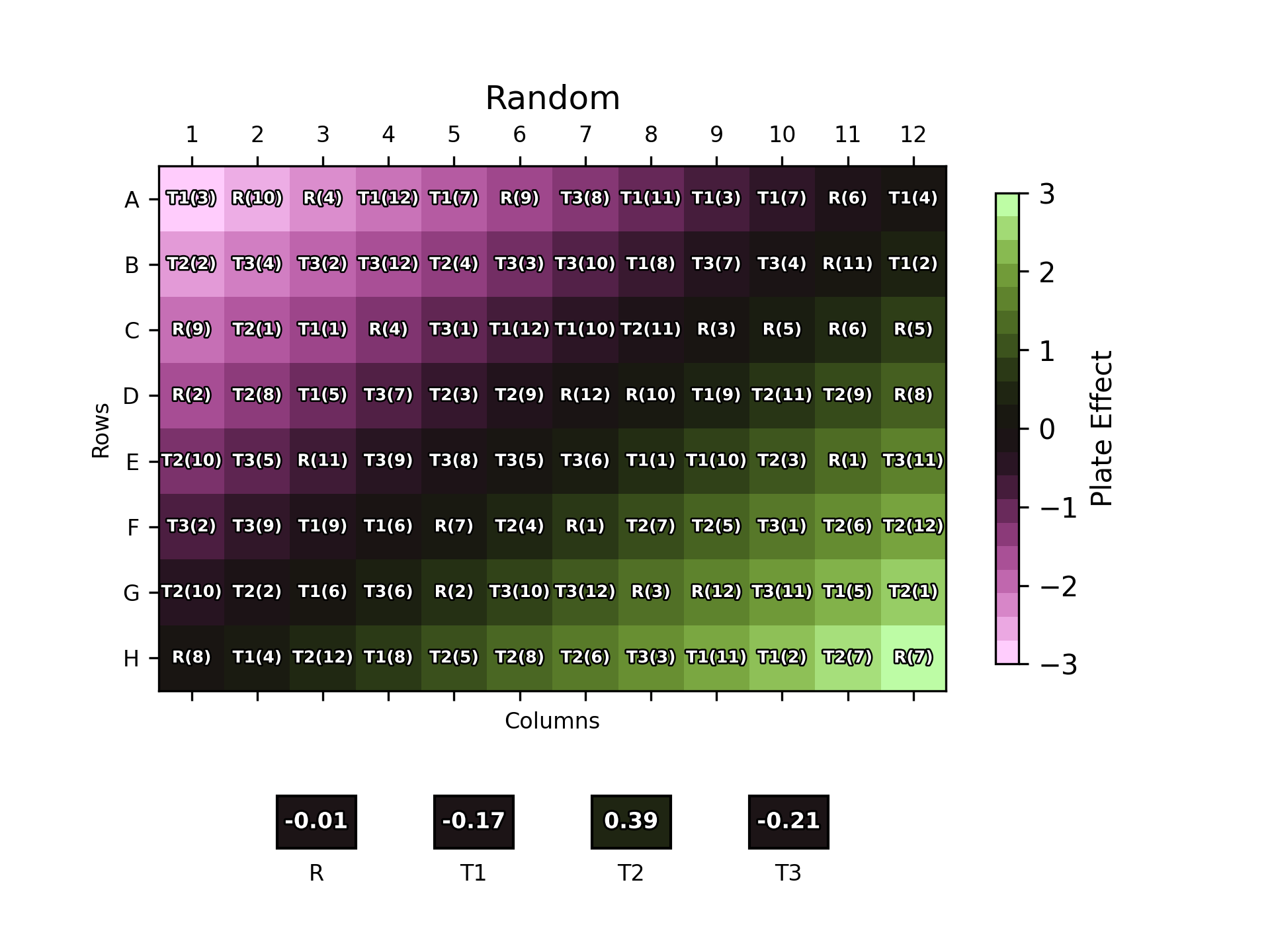
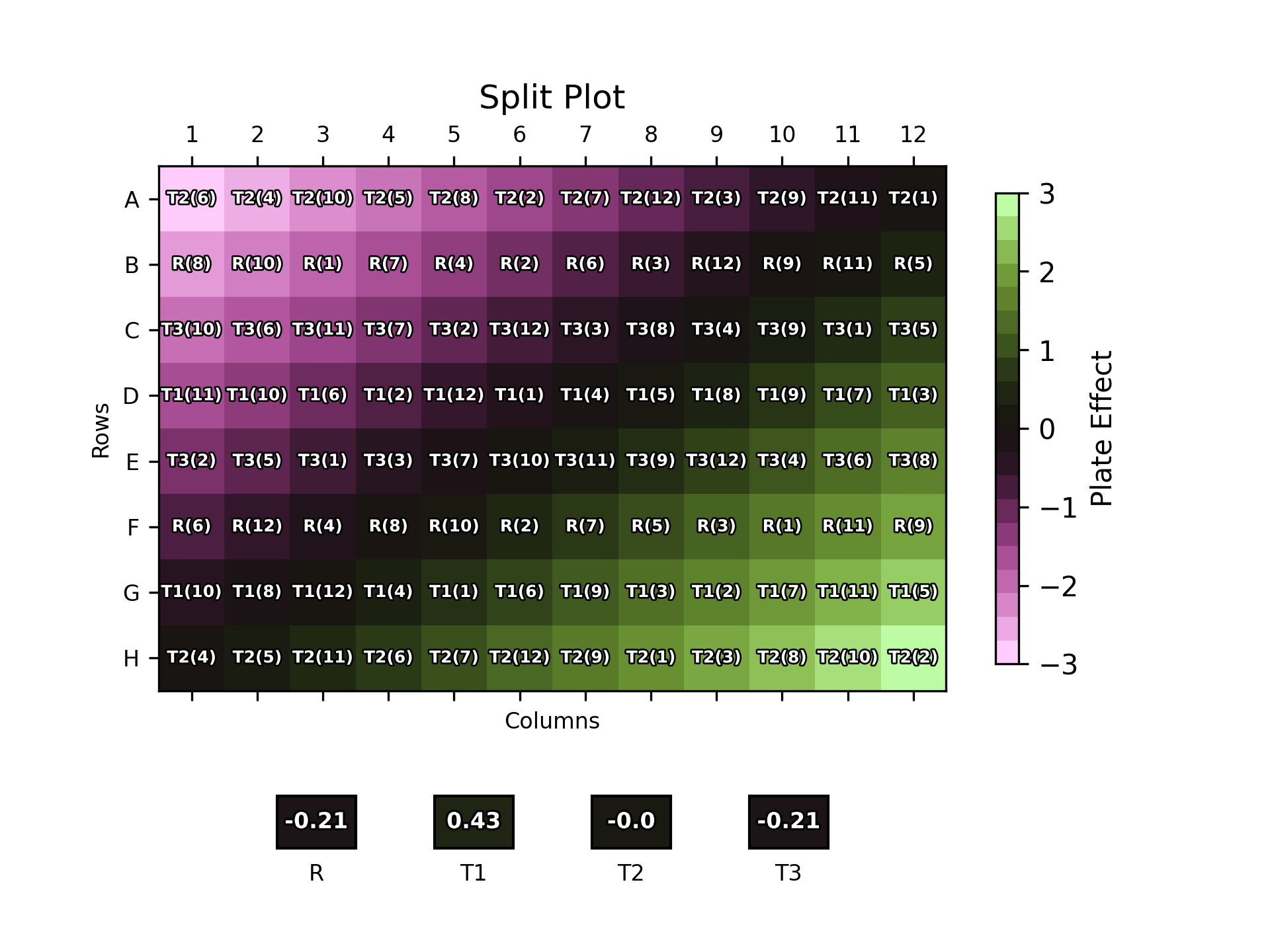
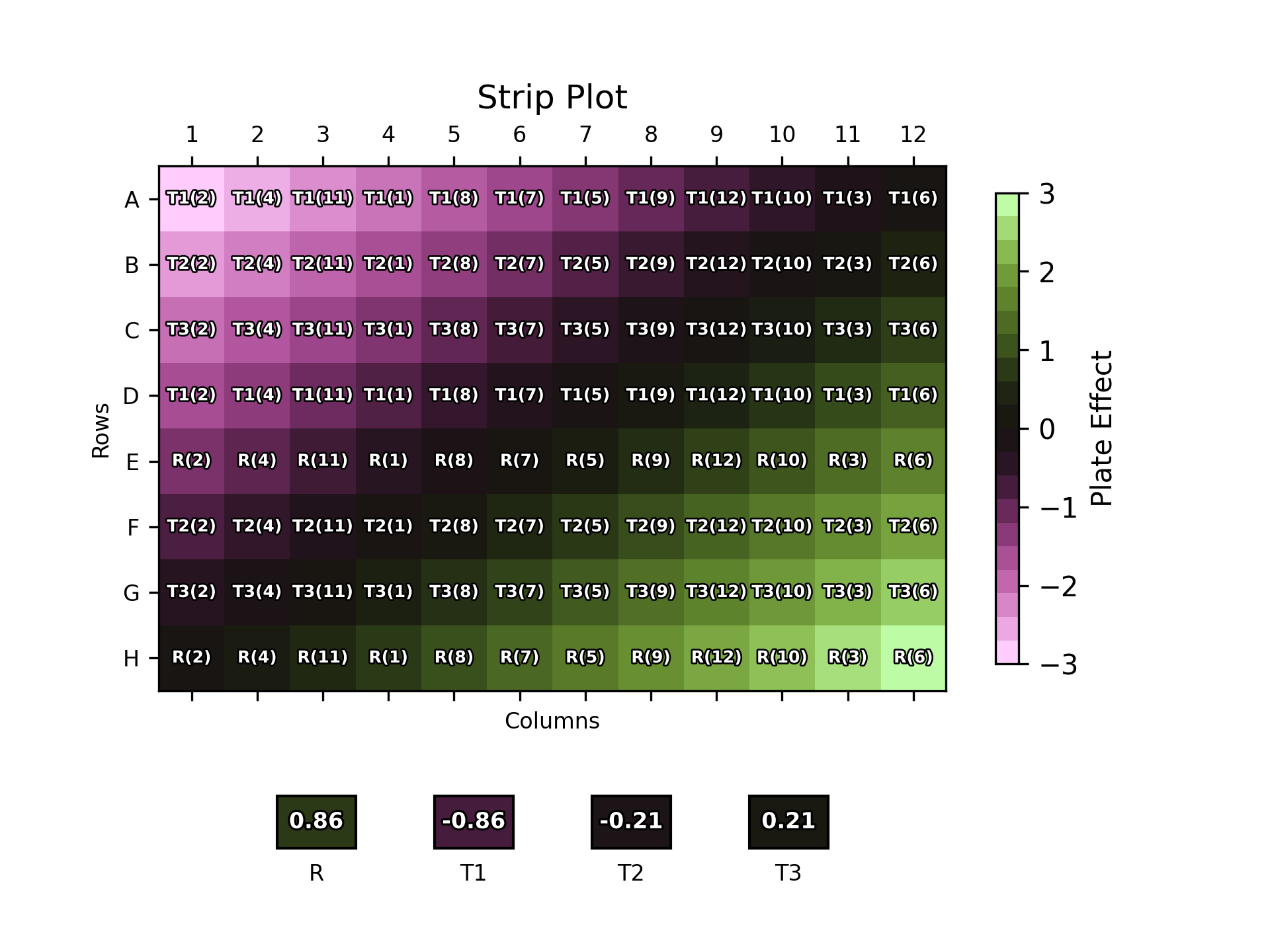
The position of each dose of each sample replicate on the plate is labelled over the plate effect heat map. The boxes below the plot show the overall plate effect each sample “experiences” through the position of its two replicates on the plate. This follows the same colour mapping as the heat map: a green or purple box indicates that the sample experiences strong plate effects, while a black box shows a more neutral environment.
As one might expect, the naïve plate layout (top left) performs poorly, with the reference and T3 samples both experiencing strong plate effects. T1 and T2, conversely, experience a weaker overall effect due to their positioning near the middle of the plate where the plate effects have least magnitude. The randomised designs – full randomisation (top right), split-plot (bottom left) and strip-plot (bottom right) perform better, with overall plate effects closer to zero for most samples.
Notably, however, this is not the case across the board: for example, T1 experiences a -0.86 overall plate effect using this particular strip-plot layout compared to -0.43 with the naïve layout. This highlights that, since we – deliberately – have no control over the position of the samples on the plate, there is nothing preventing the doses from any one sample clustering by random chance. If this happens to be in a region with strong plate effects, then this would result in a large overall plate effect for that sample.
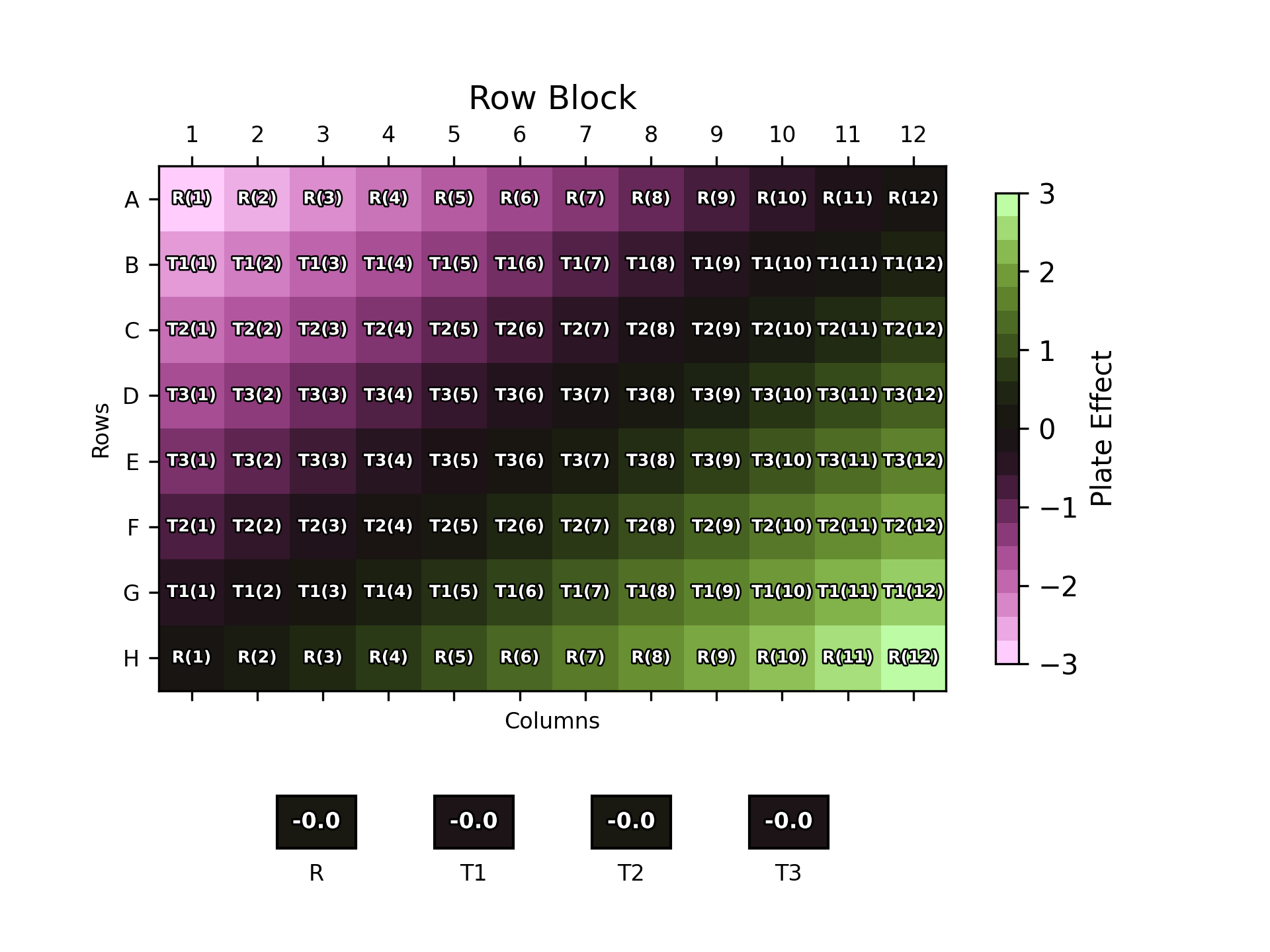
In a scenario such as this where we have a good understanding of a consistent plate effect pattern, we can minimise the overall plate effect experienced by each sample by strategic plate layout choices. The plot below shows one such example. Here, we have split the plate into two blocks – the top half and the bottom half. One replicate of each sample is included in each block and – importantly – the order of the samples is mirrored between blocks. This ensures that each sample is, on average, in the middle of the plate where the plate effects are weakest. In this simulated case, this results in an overall plate effect of zero for all four samples. Real-world data would, of course, not be quite this perfect, but this demonstrates how effective strategic choices of plate layouts can be at mitigating plate effects.
Randomisation can be built into these blocked designs if desired, as shown below. Here, we have split the plate into four 24 cell quadrants. The doses are then randomised so that three from each sample replicate are placed in each block, meaning the sample replicate experiences the plate effects associated with each quadrant evenly. This can result in smaller overall plate effects than for a fully randomised plate layouts, though as randomisation is involved this cannot be guaranteed for each individual case.
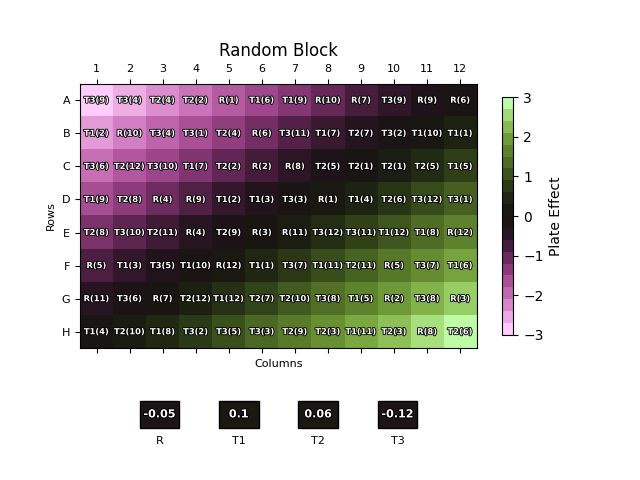
This can be taken to ever increasing levels of granularity – one could consider 2×3 blocks for randomisation, for example – but, in doing so, we again run into the trade-offs between performance, practicality, and throughput. If a more complex plate layout isn’t going to improve the performance of the assay all that much, why sacrifice the practicality? In such circumstances, a uniformity study can be extremely beneficial. An informed assessment of the accuracy and precision of the reportable result in the presence of any plate effects allows a plate layout to be chosen which achieves the desired balance between the keys to assay success.






Comments are closed.