Powerful statistical analysis at your fingertips
A Feature-Rich Statistical Analysis Package for Relative Potency and ELISA
Try QuBAS for free
The free version of QuBAS gives you the ultimate try-before-you-buy experience. Discover all of QuBAS’ powerful development features using your own data, or learn the ropes using the pre-prepared demo method.
QuBAS Now is available to use for free with no download required. Simply sign up!
Regulatory Compliance
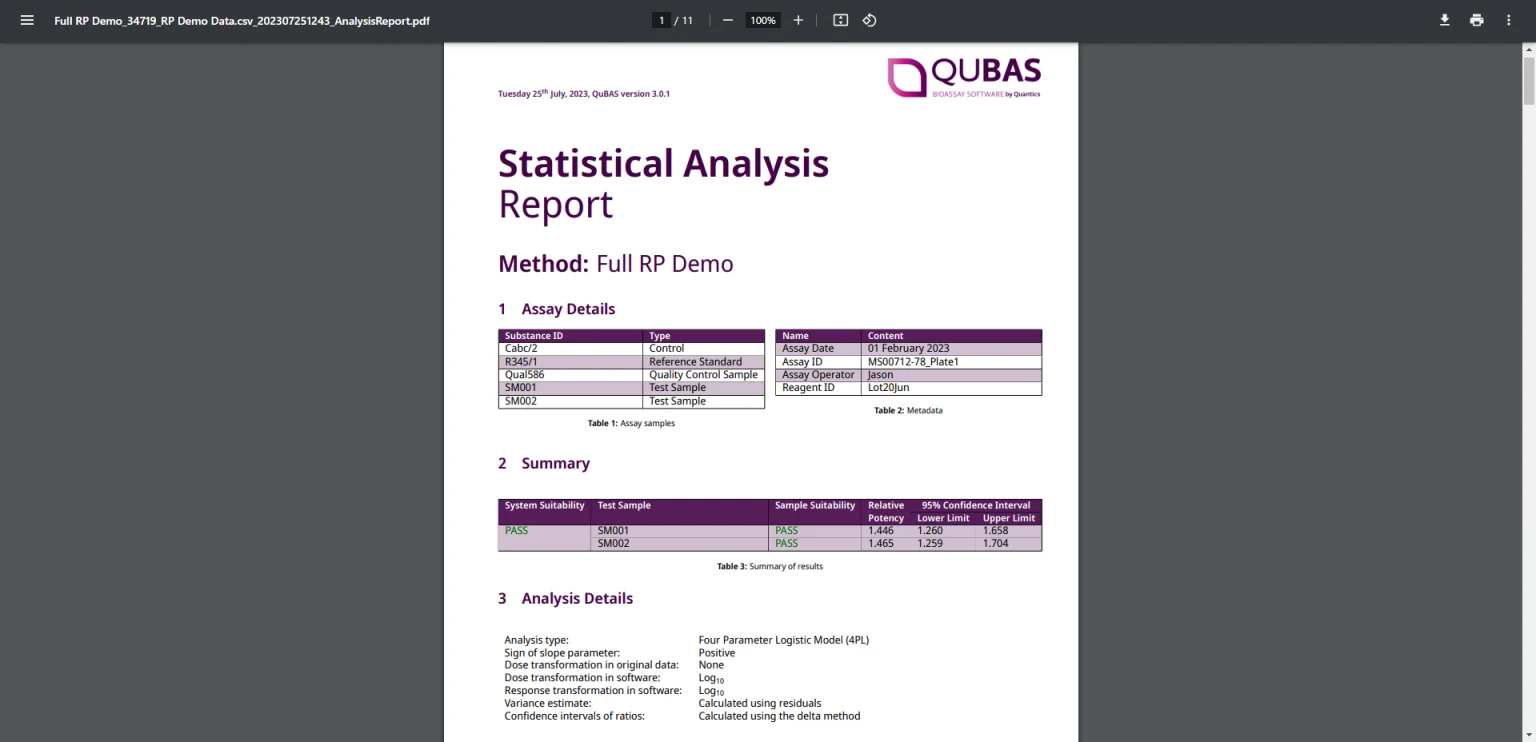
QuBAS’ unique Continuous real-time Validation (CrtV) system means that QuBAS is regulator-ready from day one. All analyses in Routine mode are 100% GMP and 21 CFR pt 11 compliant
Dedicated Support
QuBAS is backed by Quantics Biostatistics, with more than 20 years’ experience in bioassay consultancy. Whether it’s technical, user, or statistical support you require, we have a support option tailored to you.
Statistics Software for Scientists
Not Just Statisticians
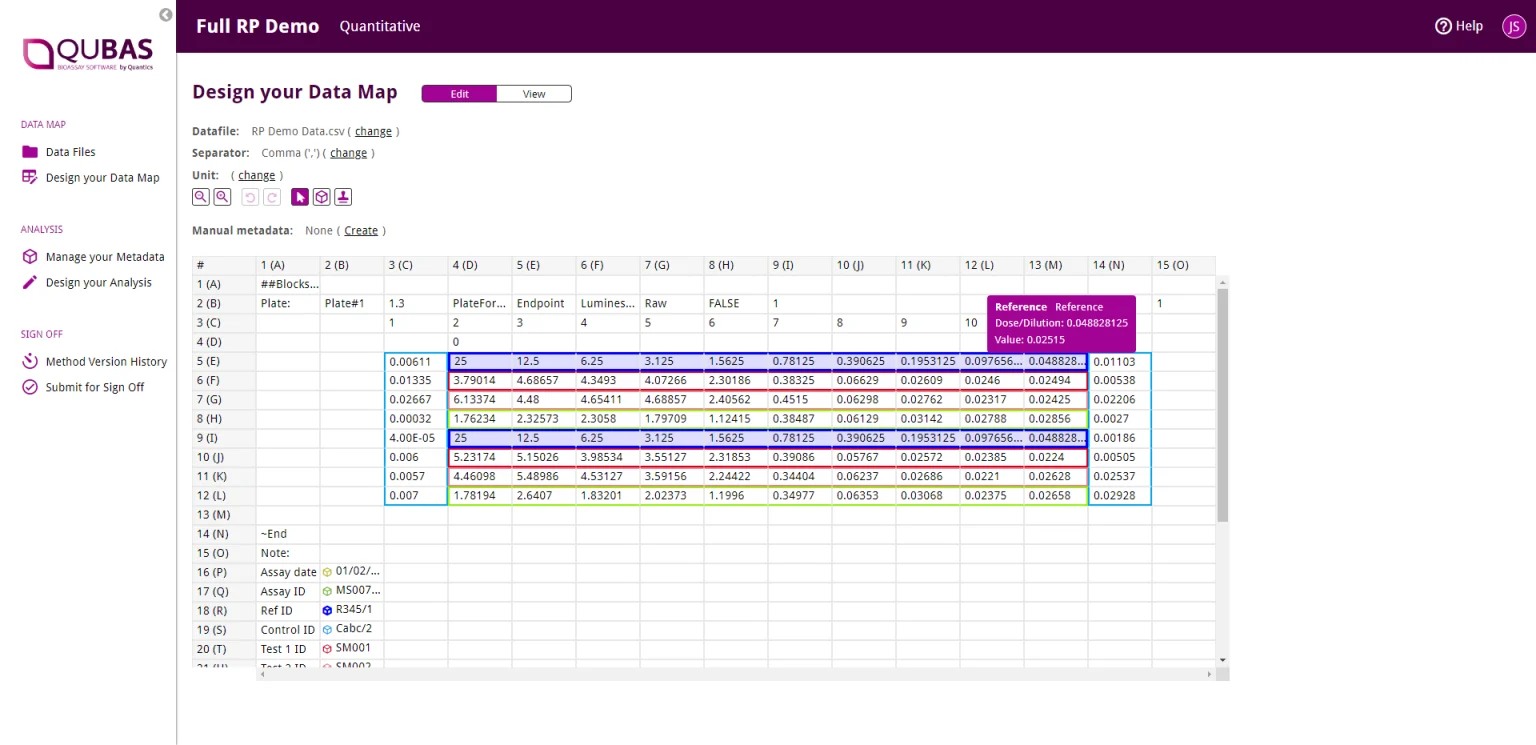
If you’re a bioassay analyst, you want to focus on running assays, not sitting through days of training just to learn how to use your statistics software. QuBAS is designed with ease of use in mind, so new users can be up to speed in minutes. We’ve built in all standard bioassay models and suitability criteria, so you can design your analysis method with the push of a button – no coding required.
Why Scientists
Choose QuBAS
Pre-Programmed Analysis Options
QuBAS comes with all USP and PhEUR standard models, analysis options, and suitability criteria pre-programmed with simple click-and-drag tools, meaning no QA/QC nightmares when preparing your analysis method.
Flexibility to Optimise your Analysis
QuBAS’ development mode includes powerful version control, allowing you to experiment with your analysis options safe in the knowledge that you can always revert to a previous version. Want to see if a whole new approach would improve your results? QuBAS gives you the flexibility to do just that without losing your current work.
Minimal training requirements
New QuBAS users can be up and running in minutes thanks to a design focused on ease of use. We know that you want to focus on what we do best, so QuBAS is specifically designed to facilitate a busy lab environment.
Improved Accuracy and Precision for ELISA Analyses
QuBAS is the only bioassay statistics package to implement the Bursa-Yellowlees method, an innovative alternative to traditional ELISA analysis techniques. By making efficient use of every single data point, QuBAS can provide superior accuracy and precision for many ELISA methods.
Regulator-Ready
Straight Out of the Box
QuBAS’ robust Routine mode is designed to enable rapid, foolproof analysis of large quantities of assay data with the powerful Automated Data Transfer functionality. With a minimalistic UI that gives easy access to all analysis results, the platform maintains a locked-down method configuration perfect for a GxP environment. Once you’ve finished your run, examine your output in the Monitoring room which automatically trends data against your choice of metadata
How QuBAS
Makes Compliance Easy
Full GMP and 21 CFR pt11 Compliance
QuBAS’ Routine mode is fully GMP and 21 CFR pt11 compliant and, with the unique Continuous real-time Validation (CrtV) system in place, QuBAS can provide regulator-ready results immediately after installation. There are no user options beyond uploading data and downloading results, so you can be certain that your analysis set-up is exactly as intended.
Seamless integration with your workflow
QuBAS’ Automated Data Transfer features can automatically import and analyse new data and export the results to a location of your choosing. After set-up is complete, you can go from a plate in an analyst’s hand to fully GMP-compliant results in the time it takes to get a cup of coffee. Better yet, it requires no user input, reducing QA/QC burden and making laborious manual data transfer obsolete.
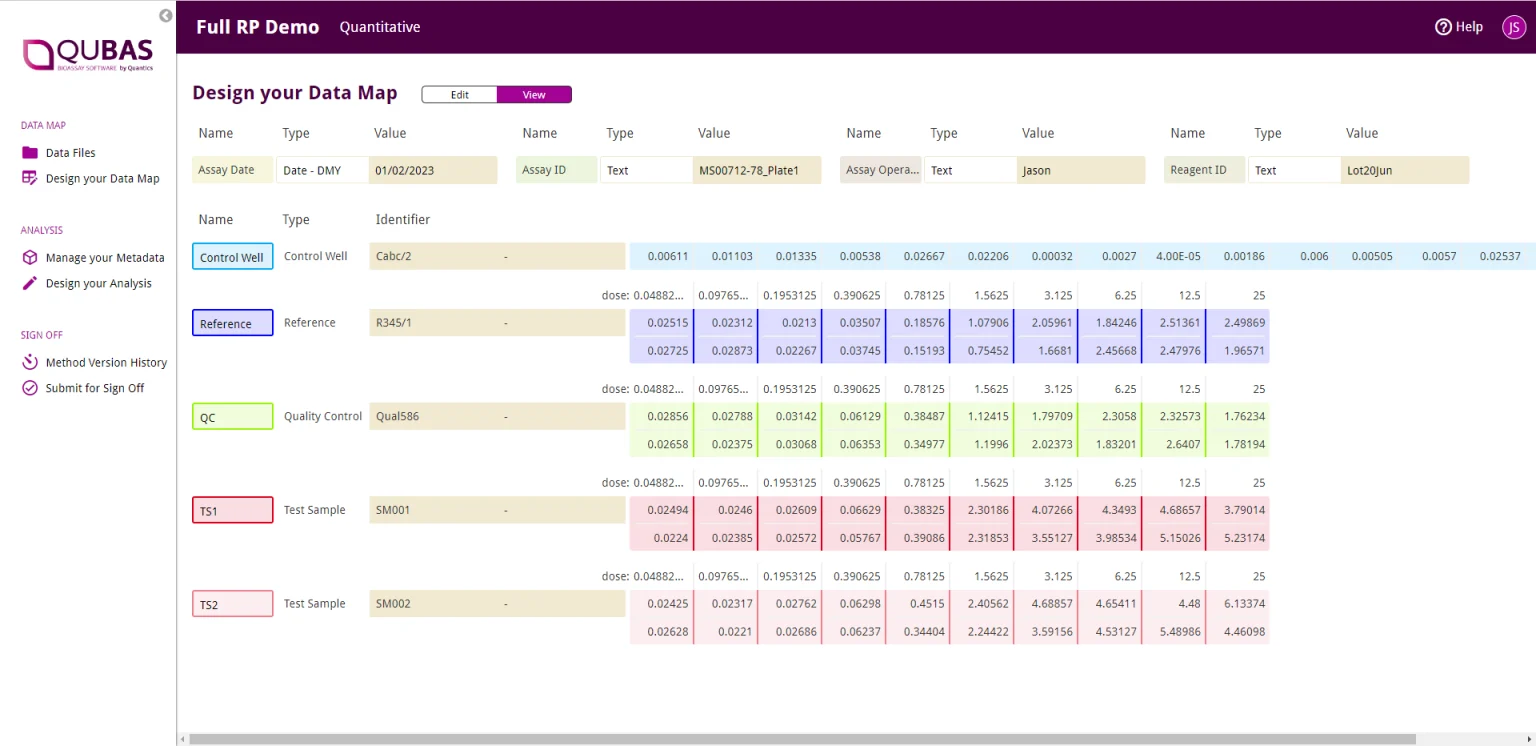
Unlimited Metadata Trending
QuBAS gives you the freedom to assign any information in your input file as metadata, which is automatically captured in the Monitoring room. Here, you can trend your results and model parameters against any designated metadata to easily keep track of your assay’s performance, helping you understand any assay failures.

Cloud-based options available (if you’d like)
QuBAS is the only bioassay software package available with a cloud-based option, giving you seamless & secure access to your analysis from anywhere in the world with minimal IT burden.
We know that cloud isn’t for everyone, which is why we continue to offer local installation options, meaning anyone can access QuBAS whether in the lab or their living room.