Statistics for
Vaccine Development
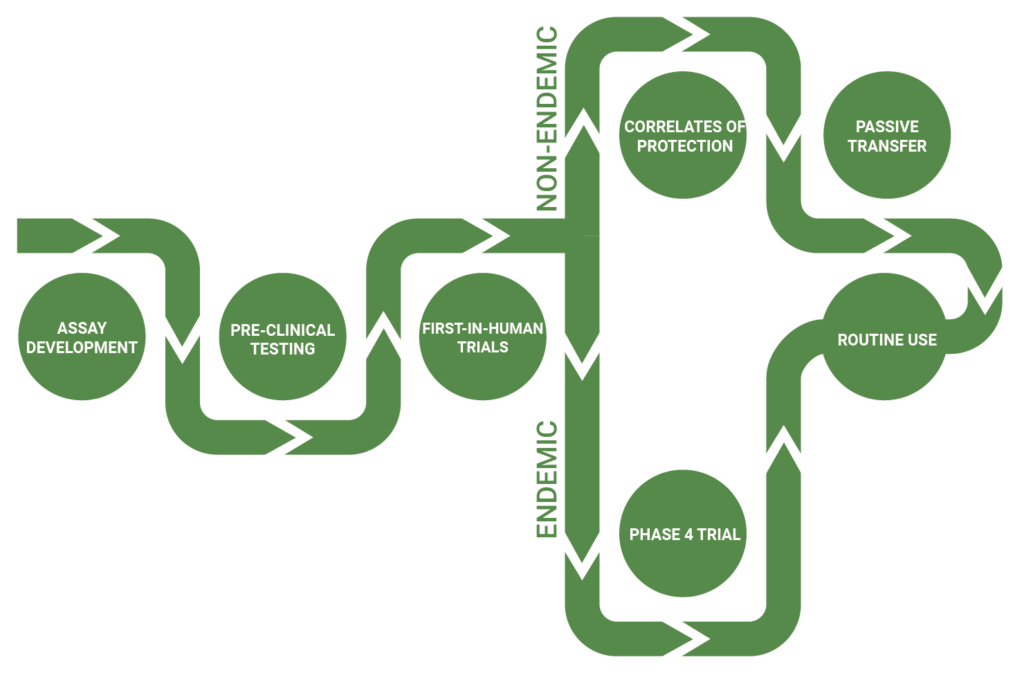
Quantics can support you through the complete vaccine development pathway
Correlates of Protection Experience
Quantics has been involved with the development, optimization and validation of a wide range of vaccine related bioassays for a number of UK, US and European customers. We have particular experience in supporting Correlation of Protection and Passive Transfer analysis for complex biodefence vaccine programs including:
Anthrax
Ricin
Bubonic Plague
Marburg
Botulinum
Ebola
Survival Analysis for Vaccine Studies
Quantics’ biomathematical experience can maximize the information obtained from expensive laboratory work. Quantics is a keen supporter of “3R” principles and our biomathematical expertise can significantly reduce the animals, replicates, and experiments required.
Many in vivo vaccine assays rely on binary data – is the subject dead or alive at a certain timepoint? Analyses which use continuous time-to-event data, such as survival analyses, are mathematically more efficient leading to improved performance. This means fewer subjects are required to achieve acceptable precision. By utilising time-to-event data instead of traditional methods, animal requirements can be reduced by >40% in some instances.
Project Insight
A number of our clients have taken survival analysis for their studies.
Client A: This client reduced the number of animals from 16 to 12 per group and improved the precision of the RP estimate.
Client B: This client was able to improve the precision of the RP estimate by a factor of approximately 2.
Client C: This client was able to improve the pass rate for their FDA-required test of parallelism (similarity) of the test samples versus the reference standard. With a probit model the failure rate was 25%; with the survival model parallelism failures were reduced to 5%.
Natural History Studies
Quantics is experienced at supporting Natural History Studies as part of a vaccine development programme. The natural behaviour of a target pathogen – including its pathology, immunology, and epidemiology – has a strong influence on the path taken during the development and testing of a new vaccine. Whether its designing studies or providing advice on endpoint selection or sample sizes, Quantics will help optimise the performance of your study.

Our Blog
Our blogs cover everything you want to know about biostatistics, from the intricacies of bioassay optimisation to the details of sample size calculations. Subscribe to make sure you never miss a thing.