Study Populations: Clinical Trial Design
The Safety Population is used for the analysis of safety, including adverse events, toxicity and laboratory evaluations. A patient should be included if, and only if, they actually received a study treatment (even if it is a placebo). This set of patients are grouped for analysis according to the treatment they actually received, as opposed to the treatment they were allocated to receive at randomisation.
Study Populations
The aim of a clinical trial is to make a calculated judgement about the likely clinical effectiveness results that would be seen if the treatments tested were to be used for all suitable patients.
Key Takeaways
- The ITT population includes all randomised patients and is usually the primary population for treatment effectiveness inferences.
- Patients should generally be analysed according to their intended (randomised) treatment, even if the wrong treatment was received.
- Different analysis goals may require different populations (PP, PK, Safety), each with clear rules.
The eligibility criteria for a patient entering a clinical trial should ensure that they are representative of patients suitable for the treatments being tested and compared.
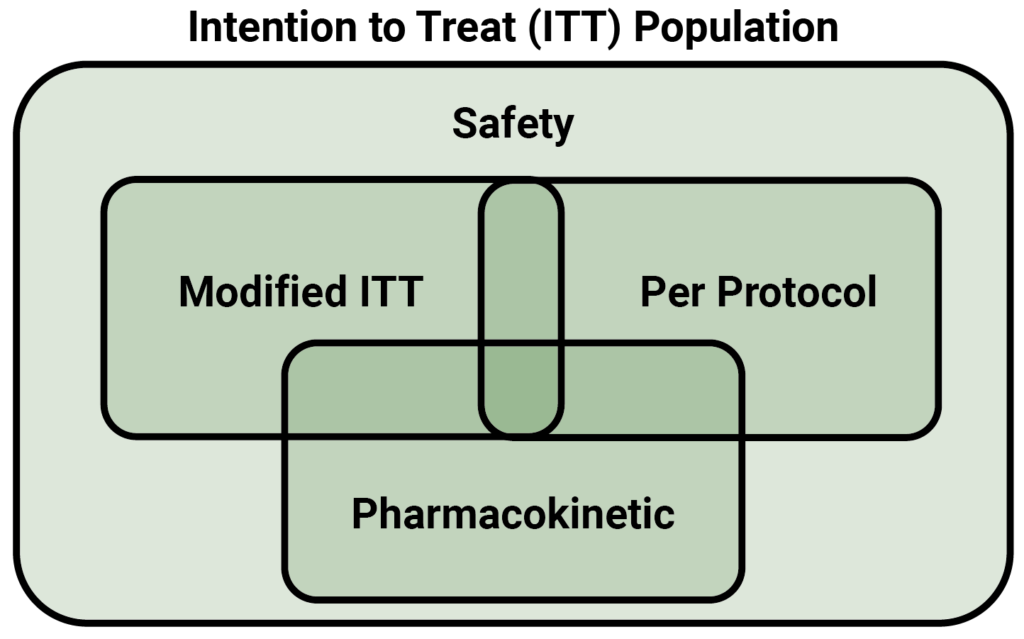
In a randomised trial, the set of all randomised patients is known as the ‘intention to treat population’, or the ITT population. This clinical trial study population is intended to represent suitable patients and to be reflective of what might be seen if the treatment was used in clinical practice. Therefore the ITT population should normally be the basis for inferences about the effectiveness of the treatments.
The eligibility criteria for the ITT population and treatments they receive will be clearly defined in the study protocol but in practice not everything goes perfectly to plan. Therefore, we need to plan ahead for the inconsistencies that can occur, for example, because of human error, and consider how we deal with them statistically.
One deviation from the protocol that is often experienced is when the allocated treatment is not actually received by all randomised patients; sometimes patients receive the wrong treatment or incomplete treatment. These things can, however, also happen to patients in clinical practice so they should not result in patients’ exclusion from the evaluation made on the ITT population. For this same reason, patients should be analysed according to their planned (intended) treatment and not the actual treatment they received. In other words, patients in the placebo treatment group should be analysed as though they received this treatment even if they received the new treatment being tested by mistake. Attempts to adjust the population groups to account for deviations can have unintended statistical consequences and are against ICH guidelines.
Another inconsistency sometimes found when analysing the study data is when the value of an endpoint for a patient is missing. This can happen when the patient withdraws from the study or is lost to follow-up. In these cases a value must be imputed in order for an ITT analysis to be conducted – otherwise, randomised patients will have been excluded and the analysis will not be ITT. The methodology for calculating these values is not within the scope of this blog but we will be covering that in our next edition.
In rare circumstances it is acceptable for a modified ITT population, or mITT population to be used in place of the true ITT population. This can be defined in various ways; often the definition is ‘all randomised patients who received study treatment’. This is discouraged by regulators, however, and a full justification is required if the full ITT is not used for the primary inferences. Further reading of the ICH Statistical Principles For Clinical Trials E9 (found here) is recommended whenever considering a modified ITT population.
Subpopulations of the ITT population are often of secondary interest. For example, the per protocol population, or PP population is usually defined as all patients completing the study without major protocol deviations – that is, those who followed the rules of the study. The PP population is normally fixed following clinical review of all protocol deviations at the end of the study; in a blinded study the clinical review must take place prior to unblinding to avoid bias.
Study populations may also be defined for specific kinds of analysis. For example, if the study involves taking a special set of blood samples for pharmacokinetic (PK) calculations, a PK population consisting of all patients who provided suitable PK samples would be used for the PK analysis. The PK population may require expert pharmacokinetic input to determine whether a patient’s PK profile is suitable to be used in the analysis of the results.
For the analysis of safety, including adverse events, toxicity and laboratory evaluations, a patient should be included if, and only if, they actually received a study treatment (even if it is a placebo). This set of patients is called the Safety population, and patients are grouped for analysis according to the treatment they actually received, as opposed to the treatment they were allocated to receive at randomisation.
Next time we will discuss missing data in more detail and explain the most appropriate statistical approaches to dealing with them.







